Research projects
Netrins regulation of epithelial plasticity in development and wound healing.
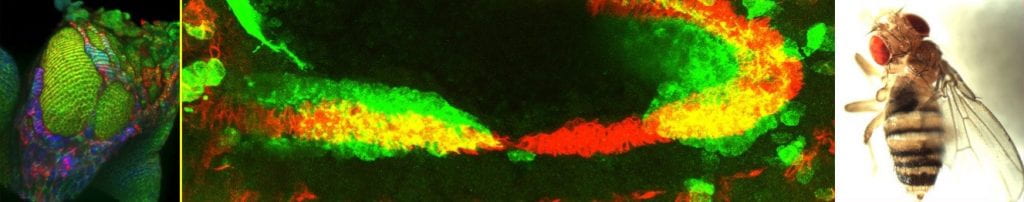
Much of the lab’s work focuses on the role of Netrins, versatile proteins best known as secreted axonal chemoattractants, but also now, strongly implicated in epithelial plasticity events (reviewed in Chaturvedi and Murray, 2021 – Cells, Tissues, Organs, In Press). We first discovered Netrins regulate epithelial cells by promoting the EMT that occurs during wing eversion (Manhire-Heath et al. 2013, Nature Communications). Surprisingly we then found Netrins also play a role in the reverse process, the MET that occurs during formation of the intestinal epithelium in the embryo. Netrins emanating from the visceral mesoderm help guide and polarise the migrating midgut cells so that they can undergo the MET (Pert et al., 2016, Biology Open). We subsequently demonstrated that loss of the Netrin receptor, Neogenin in human CRC cells induces a partial EMT (Chaturvedi et al., 2020, Scientific Reports).
Our current work is focussed on the role of Netrins in wound-healing. In collaboration with Patricia Jusuf’s lab, we’ve found that Netrin expression is rapidly upregulated at the edge of epithelial wounds, in human, zebrafish and fly and are exploring the signaling pathways involved. Thus, in both development and damage, regulation of epithelial integrity involves Netrins.
Left to Right: EMT in the peripodial epithelium; Migrating midgut cells (green) in the embryo; Eversion failure due to loss of NetA.
Epithelial homeostasis in the adult midgut.
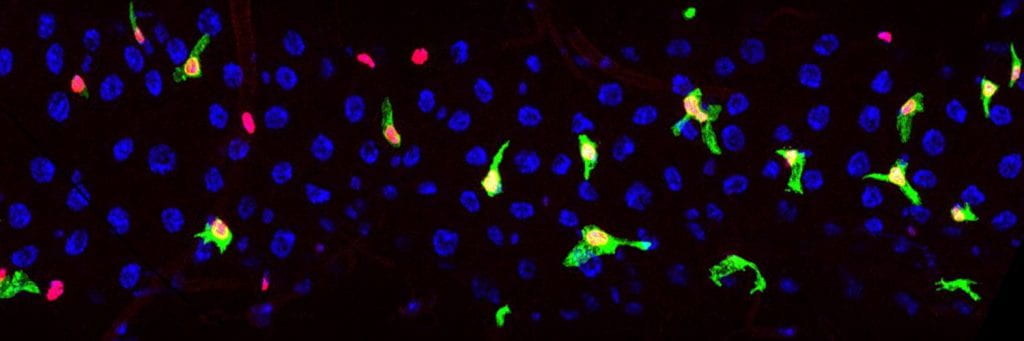
The green cells shown in the image below are enteroblasts. These are motile progenitor cells that, when required, can undergo an MET and differentiate into the absorptive enterocytes that line the gut (large blue cells). We are studying the morphogenetic mechanisms enteroblasts employ to maintain their spatial distribution and incorporate into the epithelium.
GBE-Su(H)-GAL4 driving membrane-targeted GFP and Histone-RFP labels the enteroblasts and newly differentiated enterocytes (image Fionna Zhu).